MHRA asks pharmacists for views on acne medicine risks
In News
Follow this topic
Bookmark
Record learning outcomes
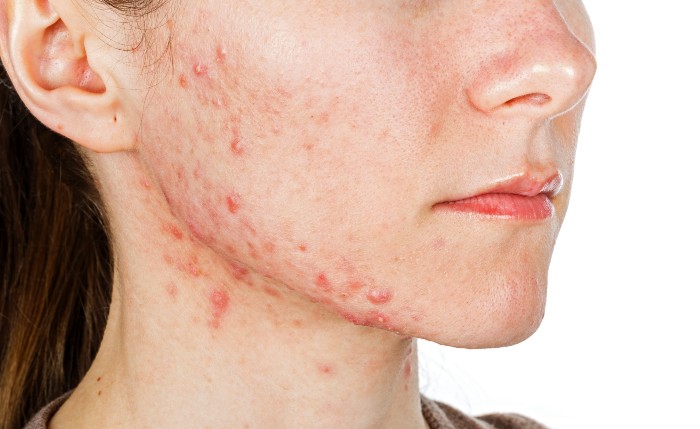
The Medicines and Healthcare products Regulatory Agency has launched a review into the possible link between the acne medicine isotretinoin and psychiatric and sexual disorders.
The regulator has asked healthcare professionals including pharmacists, as well as patients, their families and other stakeholders for their views on the risks associated with the treatment and measures that could be taken to ensure it is used safely.
The MHRA said an expert working group is looking at current evidence on isotretinoin and will inform it on what action, if any, needs to be taken including making information for patients clearer and more detailed.
“We want patients taking isotretinoin to be informed of the risks and benefits and have recently issued a reminder to healthcare professionals,” said Dr Sarah Branch, the director of vigilance and risk management of medicines at the MHRA.
“All medicines can cause side effects, and we need to ensure that the benefits of taking isotretinoin outweigh the potential risks associated with this medicine. That is why we need people’s help to gather information on psychiatric or sexual disorders, that they have suffered, and suspect to be associated with isotretinoin treatment.
“We are asking patients, family members and healthcare professionals to contribute to the review so that the Expert Working Group has-up-to-date information to consider. All information we receive will be treated confidentially.
“We know this is a personal issue for anyone who suspects they have suffered psychiatric or sexual disorders with isotretinoin treatment, therefore we would appreciate any help you may be able to give us.”
