Clinical

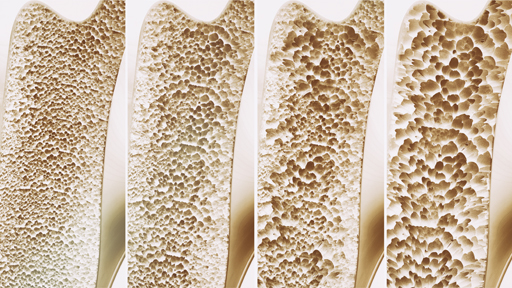
Confusion over treatments undermining osteoporosis care
In Clinical
Let’s get clinical. Follow the links below to find out more about the latest clinical insight in community pharmacy.Bookmark
Record learning outcomes
Confusion over drugs for osteoporosis is leaving some people vulnerable to potentially preventable fragility fractures, a leading charity has warned.
Key facts
- Community pharmacists can help raise patient awareness of the differences between the growing number of treatments for osteoporosis
- Prescribers need to appreciate why a ‘holiday’ might be appropriate for bisphosphonates but not for denosumab
- New drugs for osteoporosis using novel mechanisms will soon be available
The National Osteoporosis Society has seen a spike in calls to its helpline from patients reporting their denosumab treatment has been stopped. “We have also had reports from our health professional colleagues and expert clinical advisers across the UK that denosumab has been stopped inappropriately,” says Dom Hall, a NOS spokesperson.
The concerns focus on two side-effects of denosumab and bisphosphonates – atypical femoral fracture and osteonecrosis of the jaw – but both these are rare. According to the MHRA, the risk of atypical femoral fracture after at least 2.5 years of treatment with denosumab is about one in 1,000 to one in 10,000.
“The risk of these very rare side-effects seems to be linked to duration of treatment,” says Dom Hall. “Because of this, ‘how long to treat’ is a hot topic. Experts and regulatory authorities have gone some way to resolve the uncertainty by urging regular review of treatments, usually after five years, and considering pausing treatment for a few years for some patients. However, pausing treatment is relevant only for bisphosphonates.”
Out of balance
“Recently, there’s been much more focus on these very rare risks than on the benefits in terms of fracture prevention,” adds Neil Gittoes, consultant and honorary professor of endocrinology and head of the centre for endocrinology, diabetes and metabolism, University Hospitals Birmingham.
“We need to emphasise just how rare atypical fracture and osteonecrosis are and highlight the benefits of bisphosphonates and denosumab. Healthcare professionals also need to understand the pharmaco-logical differences between them. Pharmacists can help get these messages across.
“Denosumab is highly effective at preventing fractures at all sites and shows good long-term safety in studies up to 10 years,” Professor Gittoes says. The Freedom study1, for example, enrolled 7,868 women aged between 60 and 90 years with low bone mineral density (BMD). After three years, women taking denosumab were 68 per cent less likely than placebo users to develop a new vertebral fracture on x-ray (2.3 and 7.2 per cent respectively). The denosumab group was also 40 and 20 per cent less likely than placebo users to develop hip (0.7 and 1.2 per cent respectively) or non-vertebral fractures (6.5 and 8.0 per cent respectively).
“Discontinuing or delaying denosumab may be associated with increased risk of multiple vertebral fractures,” Professor Gittoes adds. For instance, a study presented at the 2016 American College of Rheumatology meeting followed 1,471 patients for at least seven months. Over this time, 5.6 per cent of patients who discontinued denosumab experienced a new vertebral fracture and 3.4 per cent experienced multiple new vertebral fractures.
The increased fracture risk after discontinuation reflects denosumab’s pharmacology. Denosumab binds reversibly to a protein called ‘receptor activator of nuclear factor-kappaB ligand’ (RANKL) and so inhibits the cells that break down bone (osteoclasts). This, in turn, decreases bone resorption, and increases BMD.
Bisphosphonates, in contrast, attach strongly to hydroxyapatite (a form of calcium), particularly on skeletal surfaces that are being resorbed. Bisphosphonates released during resorption reduce osteoclasts’ activity. The strong binding to hydroxyapatite means that bisphosphonates persist in the skeleton for many years. As a result, bisphosphonates’ benefits on resorption do not reverse rapidly – unlike denosumab.
Different modes of action
“Understanding the mode of action helps prescribers appreciate why a ‘holiday’ might be appropriate for bisphosphonates but can be counterproductive in patients receiving denosumab. Reviewing denosumab after five years’ treatment is good practice,” Professor Gittoes says, “but, if there isn’t a compelling reason to stop it, treatment should continue.”
If a healthcare professional considers ceasing denosumab, he suggests discussing switching therapy with an expert in osteoporosis management rather than stopping abruptly.
Community pharmacists can help raise awareness of the differences between the growing number of treatments for osteoporosis, Professor Gittoes says. “Bisphosphonates are the main first-line treatment for osteoporosis. The introduction of generics seems to have encouraged an impression that all bisphosphonates are the same. This isn’t necessarily the case,” he notes. Dosing regimens differ, for instance, which potentially influences compliance.
Pharmacists can also clarify the treatment pathway, he says. “Denosumab is a second-line treatment. Clinicians need to consider which agent they will use next if they stop denosumab but there is not widespread awareness of the sequence of drug use in osteoporosis. This will become more pressing when there is a greater choice of agents.”
New options
New drugs for osteoporosis using novel mechanisms should soon reach the market. Some drugs being developed act on specific pathways that remodel bone. Anabolic agents, such as teriparatide – a truncated version of parathyroid hormone – offer an alternative to drugs targeting osteoclasts. Several other anabolic agents and other drugs that augment osteoblasts, which produce new bone, are at various stages in the pipeline.
Optimal use of these therapeutic innovations, however, assumes a certain pharmacological sophistication, which pharmacists are well-placed to provide. Rather like recent advances in cancer, many new drugs for osteoporosis work through sophisticated and subtle biological mechanisms.
“These are much smarter drugs than traditional treatments for osteoporosis,” Professor Gittoes says. “They will be used first in specialist centres but, given the large number of people with osteoporosis, GPs will probably prescribe many of these relatively soon after launch.
“Pharmacists can offer a heads up to their primary care colleagues that these drugs are common and explain the risks, benefits and mode of action.”
Treatment gap
The expanding armamentarium offers the potential to reduce markedly the incidence of fragility fracture – assuming that patients can access treatment. The European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis noted recently that only about 55 per cent of eligible patients receive osteoporosis treatment in the UK. Across Europe the proportion varies from 25 per cent in Spain to about 95 per cent in Bulgaria.2
While these figures are for 2010, more recent studies confirm that the treatment gap persists. The risk of a fragility fracture rises with age – and the UK population is getting older – yet UK prescriptions for oral anti-osteoporosis drugs (AOD) have plateaued in men and decreased among women.
Researchers retrospectively identified patients aged 50 years and older between 1990 and 20123. In women, rates of first prescription of any AOD increased from 2.3 per 10,000 person years in 1990 to a peak of 173.8 in 2009. Oral AOD prescriptions then fell by 12.4 per cent between 2009 and 2012. In men, oral AOD use rose from 1.4 to 45.3 per 10,000 person years between 1990 and 2007, followed by a plateau.
The study also highlighted gaps in care. There was a 46 per cent difference in highest incidence of AOD use among white people (149.7 per 10,000 person years) compared to black women (68.6 per 10,000 person years). The rates were 41.1 and 16.8 per 10,000 person years for white and black men respectively.
Pharmacists can help bridge the treatment gap. “Pharmacists could ask anyone over the age of 50 years or so who has a limb in plaster how they broke the bone,” Professor Gittoes suggests. “If they seem to have experienced a low-trauma fracture, pharmacists could ask if they are being seen by a fracture liaison service or its equivalent. If not, pharmacists should refer the person to their GP.”
Professor Gittoes adds that pharmacists should ensure every appropriate person receives their vitamin D supplement, obtains sufficient calcium from their diet (and if not, receives a supplement) and takes exercise.
“There is strong evidence around the treatment and preventative strategies for osteoporosis,” Professor Gittoes concludes. Yet according to the NOS, half of women and a fifth of men over the age of 50 years still experience fractures, usually due to low bone strength (osteopenia).
Until healthcare professionals understand and utilise drugs using their strong evidence base, thousands of people each year will continue to endure potentially preventable, disabling and even deadly fragility fractures. Surely we can do better than this.
We need to highlight denosumab benefits
References
1. N Engl J Med 2009; 61:756-65
2. Osteoporos Int 2017; 28:2023-34
3. Bone 2017; 94:50-55